PSA - Lets explain
Pressure swing adsorption (PSA) is a technique used to separate a specific gas from a mixture of gases, under pressure using a specific absorbent material to remove mixture of gases leaving the gas required. It operates at near-ambient temperature and significantly differs from the cryogenic distillation commonly used to separate gases. Selective adsorbent materials are used as a trapping material, preferentially adsorbing the target gas species at high pressure. The process then swings to low pressure to desorb the adsorbed gas.
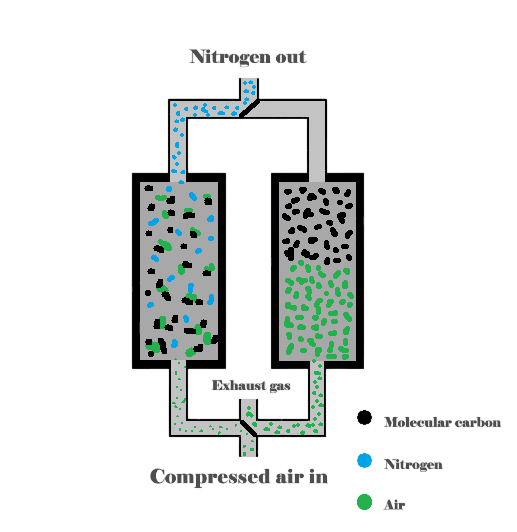
The pressure swing adsorption (PSA) process is based on that under high pressure; gases tend to be trapped onto solid surfaces (absorbed). The higher the pressure, the more gas is adsorbed. When the pressure is dropped, the gas is released, or desorbed. PSA can be used to separate gases in a mixture because different gases are adsorbed onto a given solid surface more or less strongly. For example, if a gas mixture such as compressed air is passed under pressure through a vessel containing an adsorbent bed of molecular carbon that attracts oxygen more strongly than nitrogen, the oxygen with stay on the absorbent bed and the existing gas released will be more richer in nitrogen than oxygen. When the bed reaches the limit of its capacity to adsorb oxygen, it can be regenerated by decreasing the pressure, thus releasing the adsorbed oxygen. It is then ready for another cycle of producing nitrogen from the incoming air.
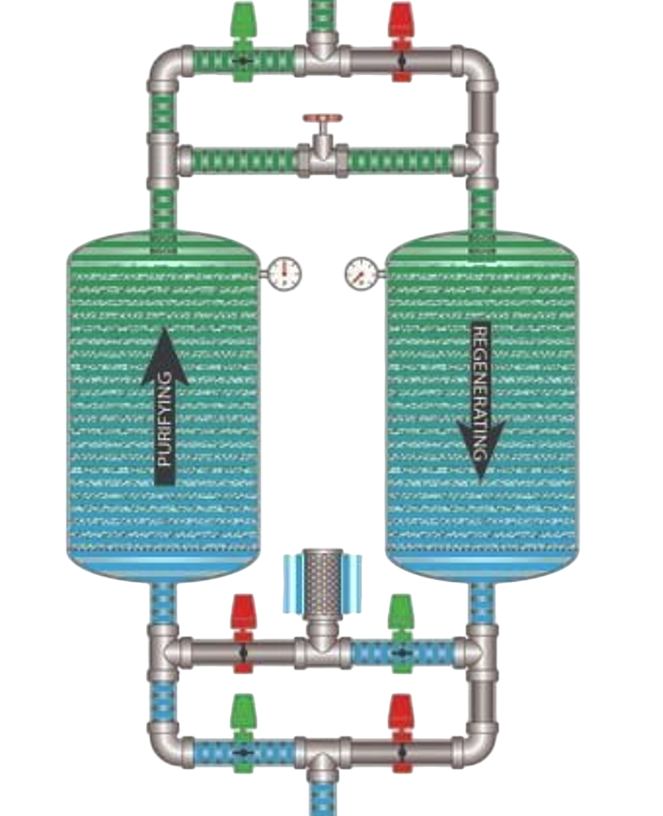
Using two adsorbent vessels allows for near-continuous production of the target gas. It also allows a pressure equalization, where the gas leaving the vessel being depressurized is used to partially pressurize the second vessel. This results in significant energy savings, and is a common industrial practice.
Process
Absorbents
Absorbents for PSA systems are chosen due to being able to discriminate between different gases, usually very porous materials chosen because of their large specific surface areas. Typical adsorbents are zeolite, activated carbon, alumina silica gel and other synthetic resins. Using materials that are of different sizes, to allow a compact fill of material, allows the surface area to be several hundred square meters, as the surface only allows a layer to be absorbed, but having small and compacted material allows more layers of the gas to collect in a large portion of the adsorbent's weight in gas.